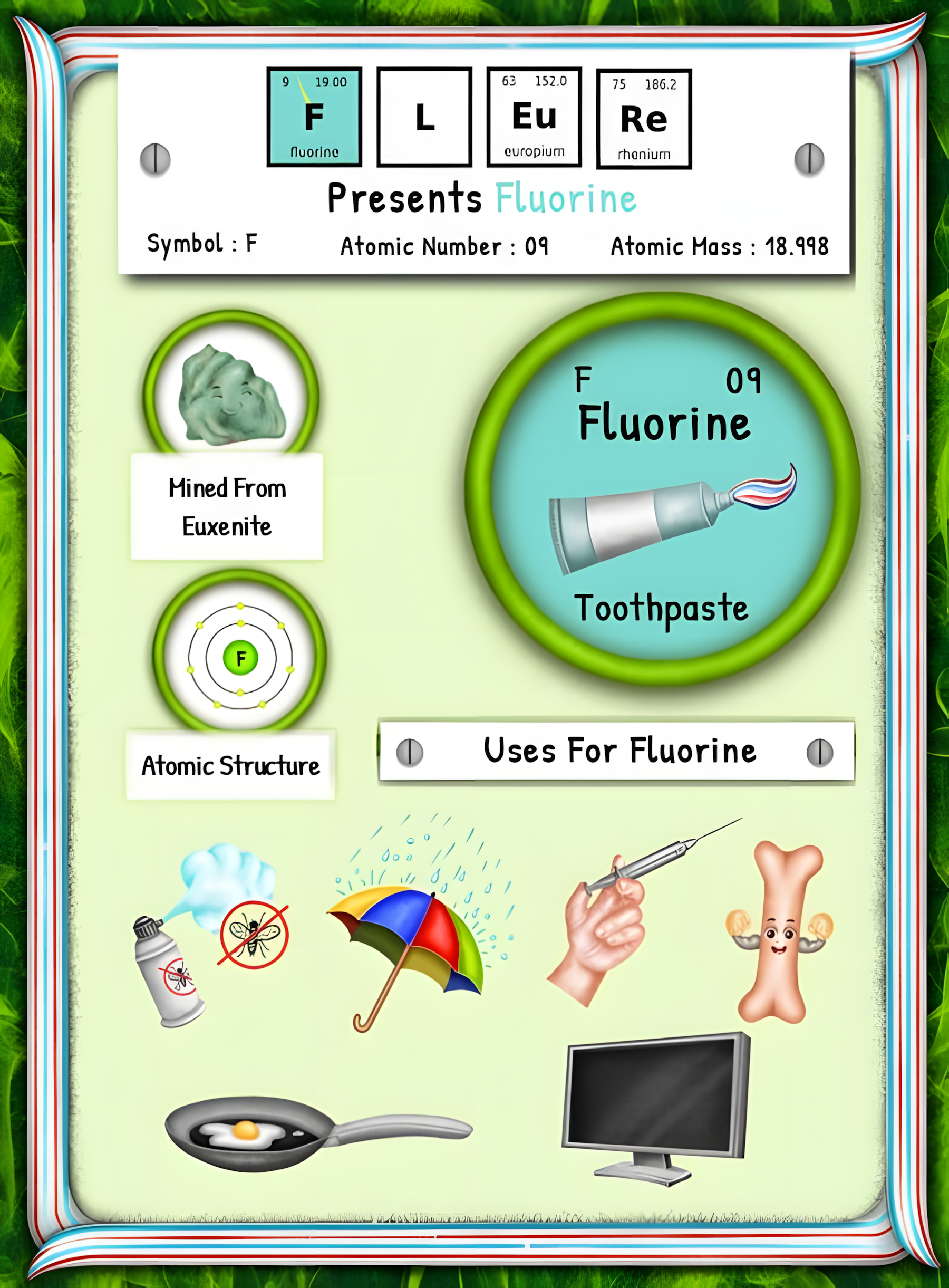
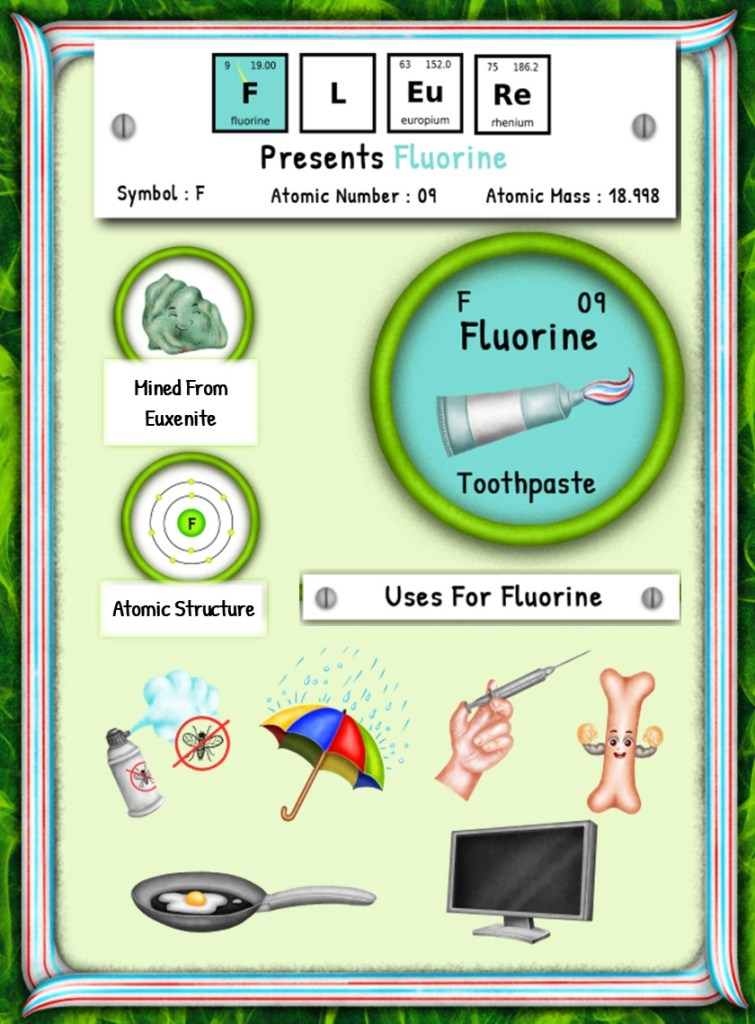
From Discovery to Disuse: The Fascinating Journey of Fluorine’s First Uses
Fluorine, the highly reactive and electronegative chemical element, was first isolated in 1886 by Henri Moissan. It quickly garnered attention for its unique properties, engendering great excitement in the scientific community. Chemists anticipated its potential applications across various industries, but over time, the impracticality and hazards associated with its use led to the abandonment of many early applications. In this article, we explore the first uses of fluorine and the reasons why it is no longer employed for those purposes today.
Early Uses:
- Metal Enrichment:
One of the initial applications of fluorine was in the enrichment of metals such as aluminum and uranium. Researchers discovered that by adding fluorine compounds to metal ores, they could extract the desired elements more efficiently. However, this process was abandoned due to the environmental and toxicological concerns posed by handling fluorine compounds. - Refrigerants and Propellants:
Fluorine’s exceptional thermal stability and resistance to chemical reactions made it an intriguing candidate for use in refrigerants and propellants during the early 20th century. However, these applications were ultimately abandoned due to the extreme reactivity of fluorine and its propensity to react explosively with many substances. The risks associated with using fluorine-based refrigerants and propellants were simply too high. - Medicinal Applications:
Fluorine’s potential as a medicinal agent was explored extensively in the mid-20th century. It was hypothesized that incorporating fluorine into drugs could enhance their efficacy and stability. Although some fluorine-containing drugs, such as the anti-inflammatory Flurbiprofen, still exist today, many early attempts at applying fluorine in medicine were abandoned due to unforeseen side effects, lack of significant therapeutic benefits, or the significant challenges associated with its synthesis.
Why Fluorine is No Longer Used:
- Extreme Reactivity:
One of the primary reasons for the abandonment of early fluorine applications is its extreme reactivity. Fluorine readily reacts with nearly all elements, leading to the potential for unpredictable and dangerous reactions. This reactivity, combined with its tendency to form highly toxic compounds, posed significant safety risks in many industrial and medical settings. - Toxicity and Environmental Concerns:
Fluorine is highly toxic, possessing the potential to cause severe health issues upon exposure. Its corrosive nature, combined with the production of toxic byproducts during certain processes, led to environmental concerns that rendered many uses of fluorine impractical. The lack of safe and efficient handling methods further limited its potential applications. - Viable Substitutes:
In many cases, other chemicals have been found to be safer and more practical alternatives to fluorine. Scientists and engineers have developed numerous compounds and technologies that offer similar or improved performance without the safety and environmental hazards associated with fluorine. This has rendered fluorine obsolete or limited to niche applications where its unique properties are still of value.
The early exploration of fluorine’s uses was fueled by the excitement surrounding its unique properties. However, limitations such as extreme reactivity, toxicity, and environmental concerns ultimately led to the abandonment of many potential applications. While fluorine continues to find use in specialized areas such as in the production of certain chemicals, its broader applications have been largely replaced by safer and more efficient alternatives. Nonetheless, the story of fluorine’s early uses serves as a reminder of the complexities involved when harnessing the power of this highly reactive element.
This article is brought to you by Sybrina Durant, the author of the middle grade picture book, Magical Elements of the Periodic Table Presented Alphabetically By The Metal Horn Unicorns. Learn More. In that book Fluorine is presented by the unicorn, Fleure. Read Fleure’s Story.

Sybrina Publishing Offers Fun Activities Based On The Book
Magical Elements of the Periodic Table Presented Alphabetically By The Metal Horn Unicorns
Browse Sybrina-Publishing on TPT or Classful or The Unicorn Periodic Table Elements Store
Inter-Active Unicorn-Themed Periodic Table from Magical Elements of the Periodic Table Presented Alphabetically by The Metal Horn Unicorns
Click here to use This Inter-Active Viewer To Learn More About The Elements Each Unicorn Represents On This Periodic Table.
Want To Hear The No Metal No Magic Song?
100 Unicorn Tee Shirt Designs – Browse To Pick Your Favorite
They are available in short sleeve tees, long sleeve tees, tank tops, sweat shirts for all ages.